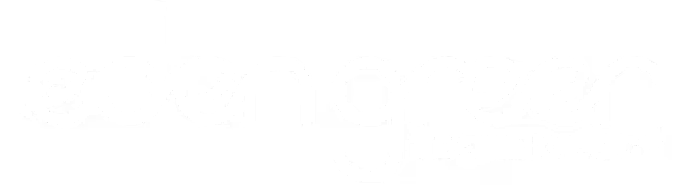Digital FSQA forms that eliminatebatch/product delayslost FSQA paperworkendless mock recallsmanual busy work
Helping FSQA teams approve batches faster, track document instantly, and stay compliant without the chaos.
book a demo.avif)





.avif)





Build dashboards to
visualize any part FSQA.
Simple FSQA forms with compliance guardrails.
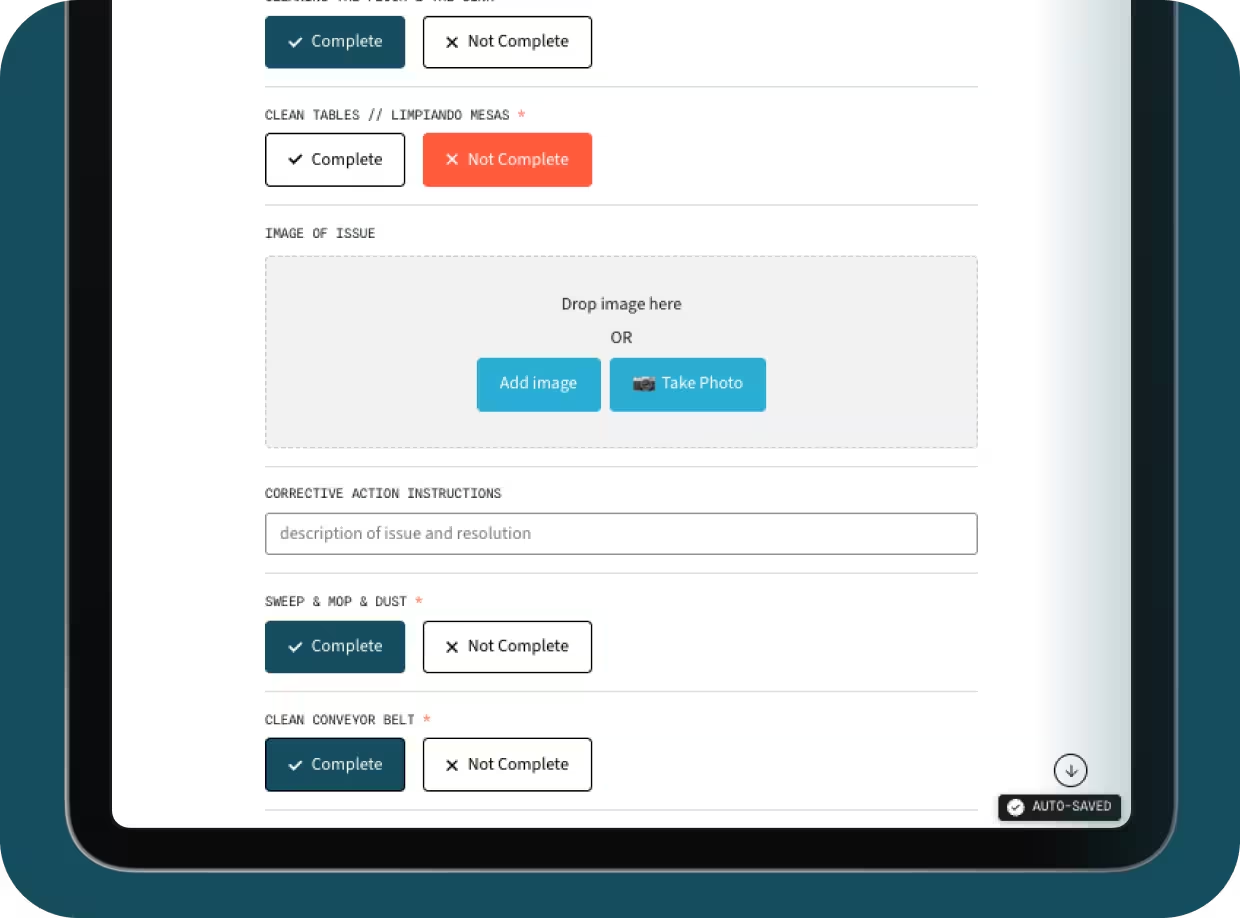
Build any custom form instantly with AI.
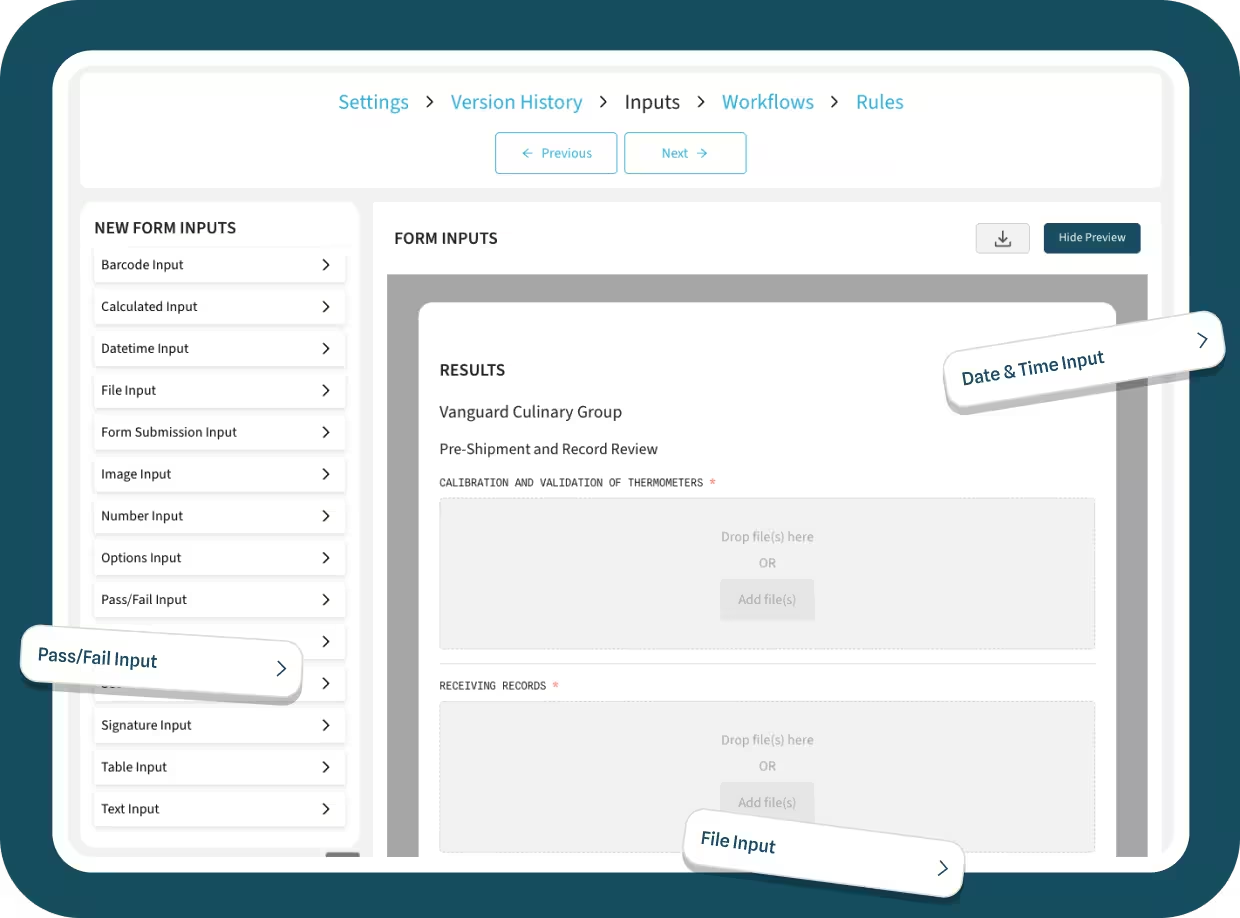
Add conditional logic and rules to any FSQA form.
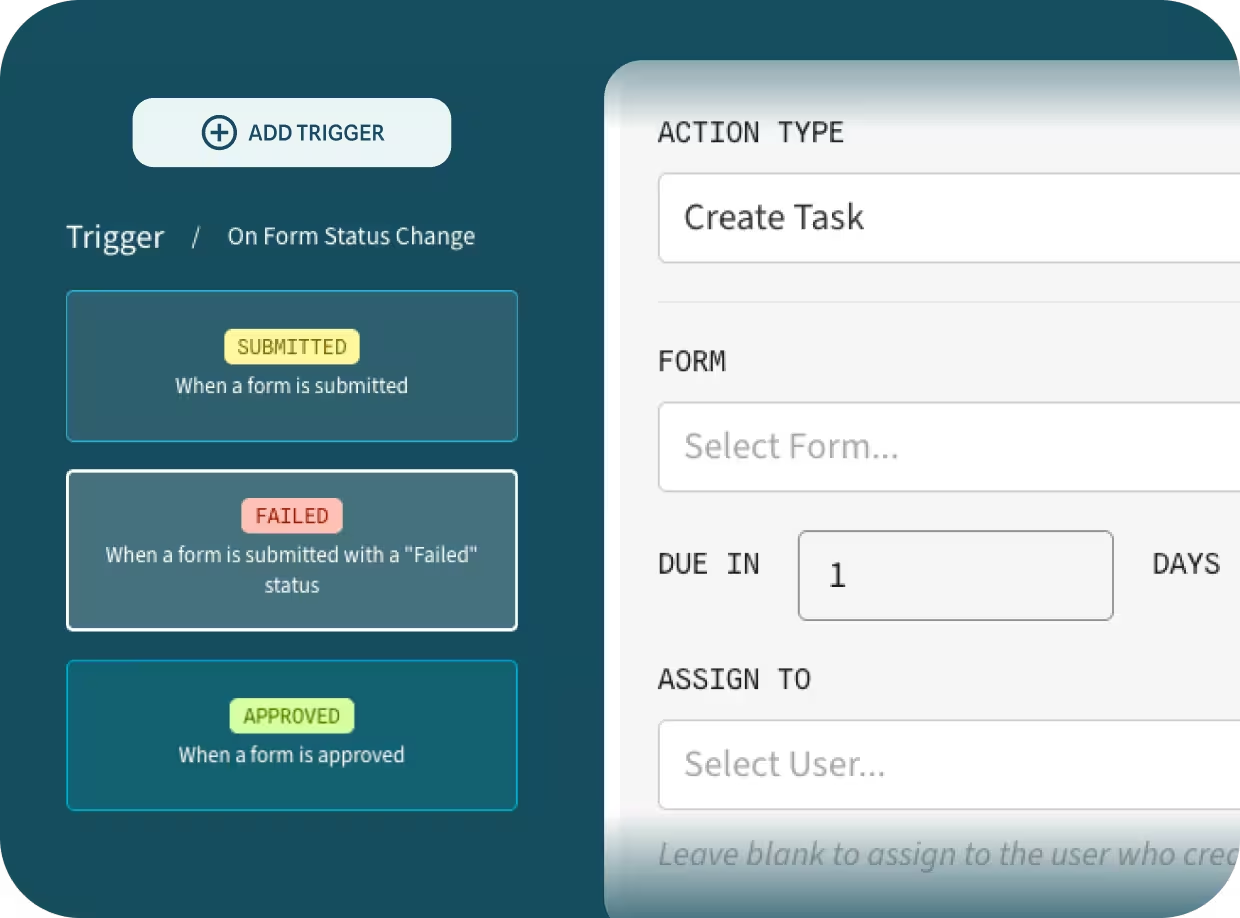
A full and traceable history of completed FSQA forms.
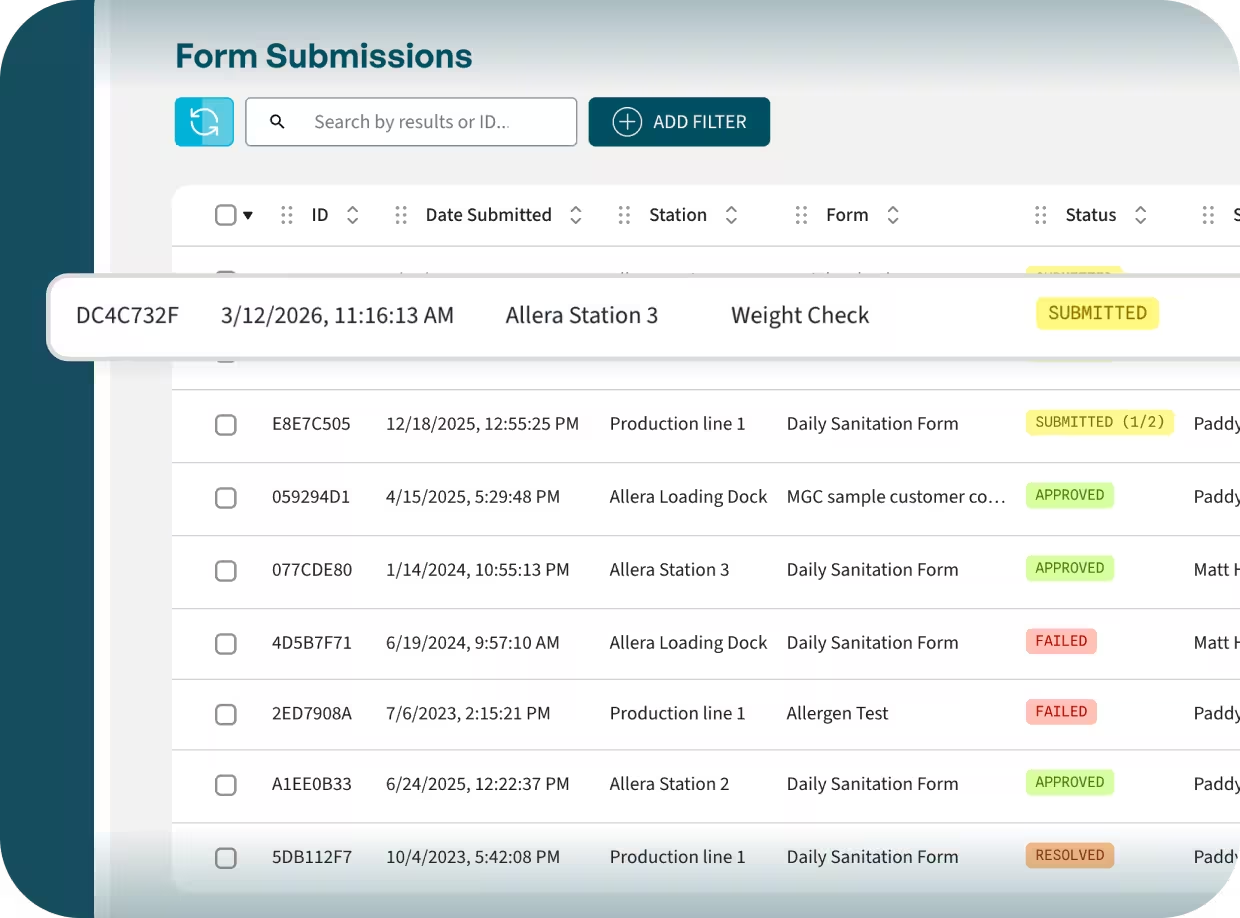
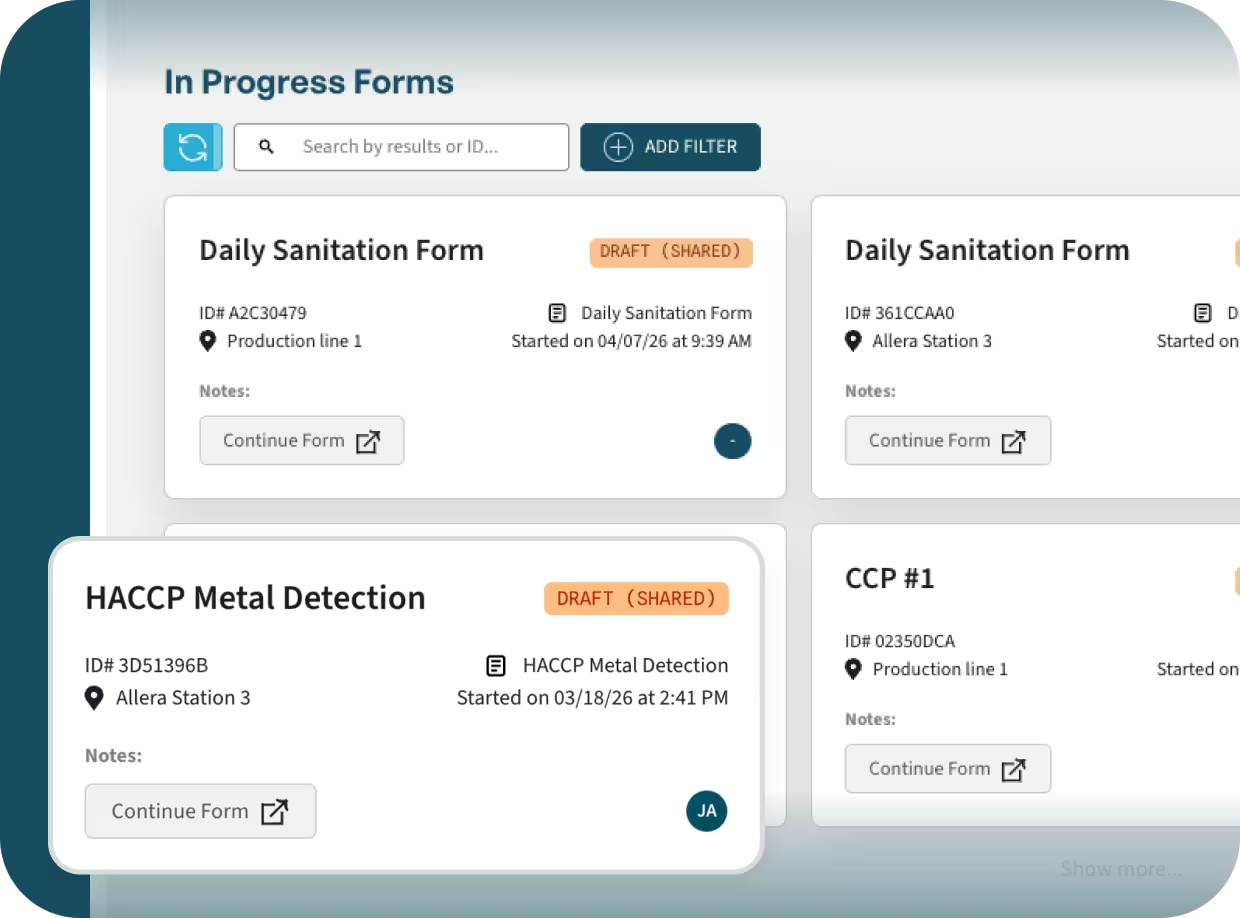
Store drafts and incomplete submissions for later.
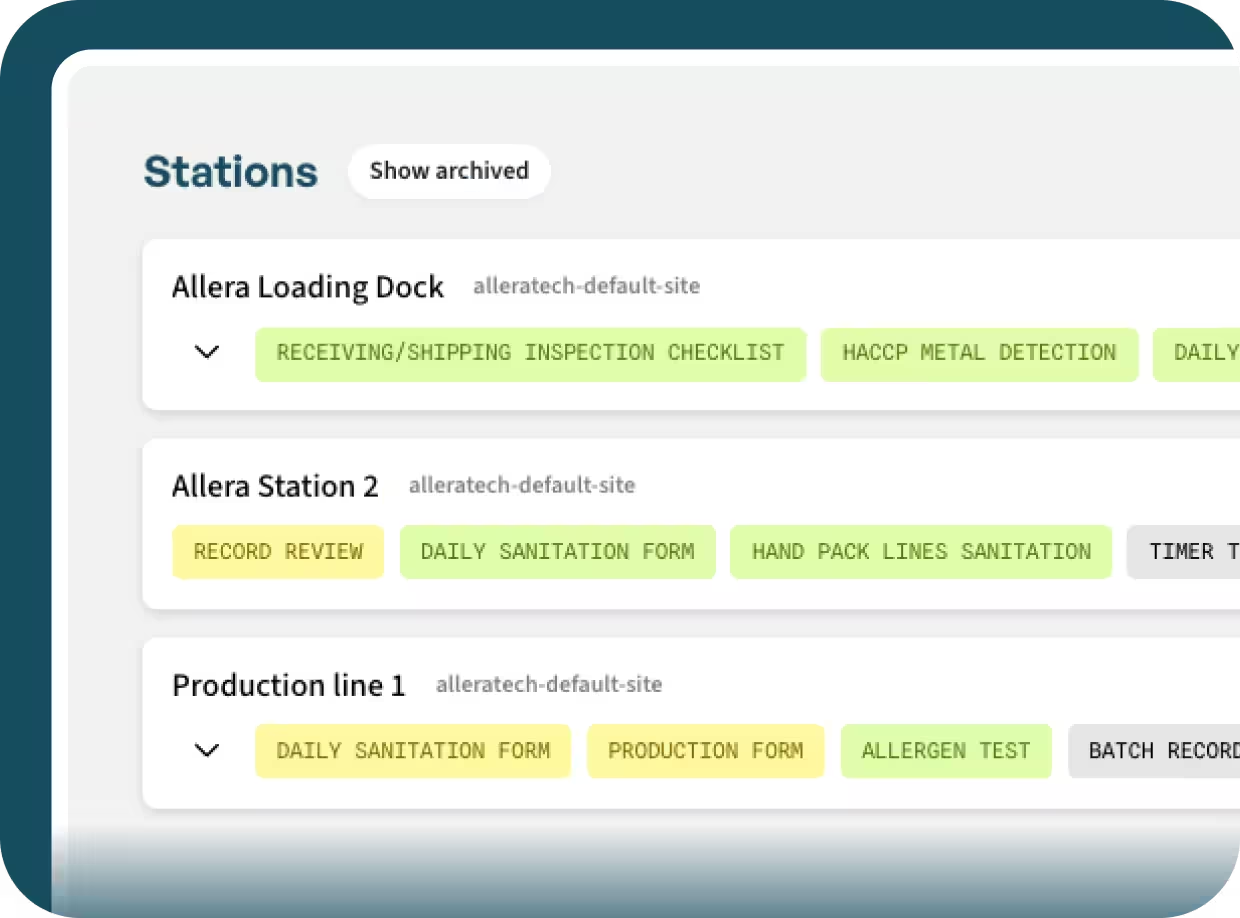
A simple way for operators to see their assigned work areas and active checklists.
Complete Document Control Platform
Everything you need to manage, control, and audit your food safety documentation in one integrated platform.
Customizable Forms
Create any form possible with the user-friendly form editor. Add failsafes, conditional logic, and automated triggers for corrective actions when deviations occur.
Time Stamps
Immediate data entry with validation checks ensures accuracy and accountability. All entries are time- and operator-stamped, making your records fully regulatory compliant and in line with FDA digital recordkeeping guidance.
Task Management
Assign QA checks and forms with due dates to operators or groups. Track completion in real time so nothing slips through the cracks — ensuring continuous compliance and smoother workflows alongside our Supplier Management Tools.
Audit Readiness
All forms are securely stored in the platform for unlimited time horizons. Search submitted/approved forms by lot code, date, form type, status, etc. Allows for quick and easy audit prep.
Verification
Streamlined workflows allow supervisors and managers to review and approve submitted forms with timestamps and user tracking. This enforces separation of duties, a key requirement for SQF Edition 10 compliance.
Traceability
Enhance food traceability across operations through receiving, processing, packaging, and shipping with lot code search-ability. Strengthen compliance with coming changes in FDA FSMA 204 traceability rules.
Analytics
Trend data with customizable dashboards for analyzing KPIs. Identify potential quality issues and make data-driven decisions, supporting continuous improvement.
User Grouping
Forms can be assigned or tagged to groups of users (e.g., Quality, Production, Line 1) to streamline workflows. Supports role-based permissions critical for compliance with GMP/cGMP guidelines.
Stories behind the results.