

8 Best Document Control Software for Food Manufacturers

Food manufacturers deal with a level of documentation complexity that general manufacturers simply don't face. Choosing the right document control software for manufacturing is one of the most consequential decisions your FSQA team will make. Your facility runs on SOPs, GMP procedures, HACCP plans, prerequisite programs, food safety plans, allergen management records, and supplier approval documentation. Each one needs to be current, approved, distributed to the right people, and instantly retrievable during an audit.
One outdated SOP distributed to the production floor is a non-conformance waiting to happen. In a BRCGS or SQF audit, document control is evaluated as a standalone element. Get it wrong and you're looking at a major finding, a corrective action plan, or worse. And in the event of a product recall, incomplete or disorganised documentation can turn a manageable situation into a regulatory nightmare. For more on the full picture of food safety recordkeeping, see our guide to food traceability.
Document control software cventralizes the creation, review, approval, versioning, distribution, and retirement of every document your facility relies on. This guide covers the 8 best document control software tools for food manufacturers, what to look for when choosing one, and how each major food safety certification scheme approaches document control requirements.
The 8 best document control software tools for food manufacturers:
- Allera: Best for food safety compliance (SQF, BRCGS, FSMA, FSSC 22000 native)
- MasterControl: Best for regulated manufacturing industries (pharma, nutraceuticals, food)
- AlisQI: Best for quality-focused food manufacturers wanting an integrated QMS
- QT9 QMS: Best for mid-market manufacturers who need predictable, all-in pricing
- ETQ Reliance: Best for large enterprises with complex multi-site quality operations
- ComplianceQuest: Best for teams already running on Salesforce
- Manufacturo: Best for shop floor document management and technical documentation
- DocuWare: Best for secure document archiving with automated routing workflows
What to Look for in Document Control Software for Food Manufacturing
Not all document control software is built with food safety in mind. A platform designed for aerospace or pharmaceutical compliance might check boxes on paper, but still leave your FSQA team doing manual workarounds to meet SQF or BRCGS requirements. Here is what to evaluate before you commit.
Version Control and Approval Workflows
Version control is the foundation of document control. Every time an SOP changes, the system should create a new version, archive the old one, and lock it from active distribution. Outdated SOPs reaching the production floor are one of the most common non-conformance triggers in BRCGS and SQF audits.
Look for automated versioning, role-based approval chains, and distribution confirmations. The software should enforce a review and sign-off process before a new version goes live. If a supervisor can bypass the approval step, the system is not really controlling documents.
Audit Trail and Electronic Signatures
Food safety auditors want to see exactly who reviewed a document, when, and what was approved. A tamper-evident audit trail satisfies this requirement and protects you during both internal and third-party audits.
If your facility is subject to FDA oversight, FDA 21 CFR Part 11 guidance on electronic records sets specific requirements for electronic signatures, audit logs, and access controls. Any document control software used in a regulated food environment should meet these standards.
Standards Alignment (SQF, BRCGS, FSSC 22000, FSMA)
GFSI-recognized certification programme owners all require documented document control systems as a condition of certification. The difference between a generic document management tool and food-safety-specific software is whether the platform understands the clause-level requirements of your scheme.
Software that maps directly to scheme clauses reduces revision time and makes gap analysis faster. When SQF Edition 10 introduced updated document control requirements, facilities using purpose-built software were alerted to gaps automatically. Facilities using shared drives were not.
Training Management Integration
Document control does not end when you publish a revised SOP. Your team needs to know about the change and acknowledge they have read it. Without a training management integration, you are manually tracking who received what version and hoping it reaches the right people before the audit.
Look for systems where a document revision automatically triggers a training assignment. Employees should sign off on the new version before it goes live in their area. This closes the loop that most document management tools leave open. For a deeper look at how audits assess this, see our guide to food safety audits.
Multi-Site and Role-Based Access
If you operate more than one facility, your document control software needs to handle centralized control with site-level visibility. Corporate can push a master SOP to all sites, but site managers should only see what is relevant to their location and role.
Role-based access also ensures that operators on the production floor see only current, approved documents: no drafts, no archived versions, no documents from other departments. This is an audit requirement under most GFSI schemes, not just a usability preference.
Quick Comparison: 8 Document Control Software Tools for Food Manufacturers
The table below gives you a high-level look at the 8 tools covered in this guide. Each one is covered in detail in the sections that follow.
The 8 Best Document Control Software Tools for Food Manufacturers
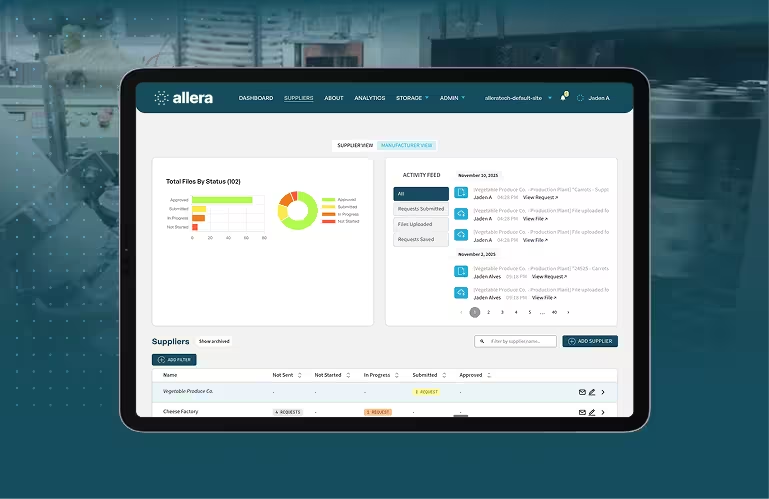
1. Allera: Best for Food Safety Compliance
Allera is the only document control platform on this list built exclusively for food manufacturers. While other tools adapt general compliance frameworks to food safety, Allera is natively aligned to SQF, BRCGS, FSSC 22000, and FSMA from the ground up.
The platform's standout feature is AI-powered compliance gap detection. When you upload or modify a document, Allera cross-references it against the specific clause requirements of your certification scheme and flags anything that does not meet the standard. When SQF Edition 10 was released, Allera surfaced gaps in existing SOPs automatically, with no manual review required. This capability is covered in more depth in our piece on AI-powered document control.
More than 500 food companies use Allera's document control software to manage their documentation, with an average implementation time of 30 days. For a platform built around the complexity of food safety certification, that is a fast time to value.
Key Features:
- AI-powered compliance gap detection that automatically flags SOPs not meeting SQF requirements
- Automated version control with time-stamped audit trails for every document change and approval
- Role-based access control and digital signatures for compliant approval workflows
- Training record integration so employees sign off on revised SOPs before the new version goes live
- Centralised document repository with scheme-specific tagging (SQF, BRCGS, FDA, USDA, etc)
Best For: Food and beverage manufacturers who need document control built around food safety certification, not adapted from a general QMS.
Pricing: From $100/month (pilot); custom enterprise pricing available.
2. MasterControl: Best for Regulated Manufacturing Industries
MasterControl is one of the most recognized compliance platforms in regulated manufacturing, with a strong footprint across pharmaceutical, medical device, and food manufacturing environments. It holds the top organic position for this keyword for good reason: the platform is mature, well-documented, and widely trusted by enterprise compliance teams.
Its document control module handles automated routing, review cycles, and approval workflows, with tamper-evident audit trails built to meet FDA 21 CFR Part 11 requirements. For food manufacturers that also operate in pharma, nutraceuticals, or dietary supplements, MasterControl provides a unified platform that eliminates the need for separate compliance systems across business units.
The main tradeoff is cost and implementation complexity. MasterControl is an enterprise platform priced accordingly, and full deployment typically takes several months. It is a strong fit for large organisations with dedicated IT and compliance resources, but may be more than a mid-sized food manufacturer needs.
Key Features:
- Automated document routing, review, and approval workflows
- Tamper-evident audit trails compliant with FDA 21 CFR Part 11
- Training management connected to document lifecycle events
- Integration with ERP systems including SAP and Oracle
- Analytics dashboards for quality KPIs and document aging
Best For: Enterprise food manufacturers that also operate in other regulated industries (pharma, nutraceuticals, dietary supplements) and need a unified compliance platform.
Pricing: Custom. Contact MasterControl for a quote.
3. AlisQI: Best for Quality-Focused Food Manufacturers
AlisQI positions itself as a quality platform first, with document control embedded within a broader QMS rather than offered as a standalone module. It is popular in European food manufacturing and ranks sixth organically for this keyword, which reflects real adoption in quality-focused food facilities.
The platform's strength is in linking document control to quality events. When a deviation or corrective action is logged, the associated procedure documents are surfaced automatically. Approval workflows are permission-based by department and role, and archived versions are retained with full rollback capability. For quality teams that want document control to be part of how they manage quality, not just a storage function, AlisQI's integrated approach is worth a close look.
The gap for food manufacturers with heavy certification requirements is that AlisQI is not specifically designed around GFSI scheme structures. It covers ISO 9001, HACCP, and BRCGS at a surface level, but does not provide the clause-level mapping that food-safety-native platforms offer. Manufacturers running best food quality management software evaluations will want to weigh that tradeoff carefully.
Key Features:
- Automatic version control with archiving and rollback capability
- Built-in audit trails linked to quality event records (deviations, CAPAs)
- Permission management and approval workflows by department and role
- Interactive process flowcharts embedded in document records
- Activity dashboards showing document status across sites
Best For: Mid-to-large food manufacturers that want document control embedded within a broader quality management platform.
Pricing: Custom. AlisQI offers demos and tailored pricing.
4. QT9 QMS: Best for Mid-Market Food Manufacturers
QT9 QMS is one of the few document control platforms on this list with transparent pricing, which makes it easier to evaluate for budget-conscious mid-market manufacturers. It appears in Google's AI Overview for this keyword and has built a solid reputation for delivering an integrated QMS at a predictable price point.
The platform includes 25+ connected quality modules: CAPA, audits, supplier management, non-conformance reports, and more, all integrated with document control. When a document is revised, employees assigned to that document are automatically notified and prompted to acknowledge the change. This training-document link is something many standalone document management tools do not offer.
QT9 is cloud-based with mobile access, which matters for production floor teams who need to pull up a procedure on a tablet without going through a shared computer. For smaller and mid-sized food facilities that want a full food quality management system without per-module licensing fees, QT9 offers strong value.
Key Features:
- Centralised document library with version tracking and electronic approval
- Automated reminders for document reviews and renewals
- Linked training assignments so employees are notified when documents they use are updated
- 25+ connected quality modules (CAPA, audits, supplier management, NCRs)
- Cloud-based with mobile access for production floor teams
Best For: Smaller and mid-sized food manufacturers that want an integrated QMS at a predictable price, without per-module licensing fees.
Pricing: From approximately $6,000/year. Contact QT9 for current pricing.
5. ETQ Reliance: Best for Enterprise Quality Management
ETQ Reliance is a highly configurable enterprise quality management platform with document control as one of its core modules. It is used by large manufacturers across food, consumer goods, chemical, and aerospace industries, and it is designed to handle the scale and complexity that comes with global operations.
The platform's defining characteristic is configurability. Approval chains, escalation paths, notification rules, and reporting dashboards can all be built to match your organisation's specific processes. That flexibility is genuinely useful for large food manufacturers with complex multi-site structures, but it comes with a corresponding implementation investment. ETQ is not a plug-and-play solution, and organisations without dedicated IT and quality systems resources will feel that during deployment.
For food manufacturers with the infrastructure to support it, ETQ Reliance provides the depth and control that simpler platforms cannot match. The document lifecycle management covers everything from creation through to obsolescence, with change management linked to document revisions across the entire system.
Key Features:
- Highly configurable workflow builder for custom approval and escalation paths
- Full document lifecycle management from creation through obsolescence
- Advanced reporting and compliance dashboards across global sites
- Integration with SAP, Oracle, Microsoft 365, and other enterprise systems
- Built-in change management linked to document revisions
Best For: Large, multi-site food and consumer goods manufacturers that need a deeply configurable quality platform and have IT resources to implement it.
Pricing: Custom enterprise pricing. Contact ETQ for a quote.
6. ComplianceQuest: Best for Salesforce-Based Teams
ComplianceQuest is built natively on the Salesforce platform, which makes it a natural fit for food manufacturers and CPG companies that already run their operations on Salesforce. It appears in Google's AI Overview for this keyword alongside MasterControl and QT9, and it has gained traction in the food and consumer goods space.
Because ComplianceQuest lives inside Salesforce, there is no separate integration to maintain. Document control workflows, approval queues, and audit logs are all visible within the same environment your team already uses. AI-assisted quality analytics and risk scoring come as part of the package, which helps quality managers identify problem areas before they become audit findings.
The constraint is clear: ComplianceQuest's core advantage is tied entirely to Salesforce adoption. If your facility does not run on Salesforce, the platform's primary selling point disappears. For organisations that do, it is one of the more seamless ways to add quality management without introducing another disconnected system.
Key Features:
- Built natively on Salesforce with no integration required for Salesforce users
- Document control with automated workflows, e-signatures, and audit logs
- AI-assisted quality analytics and risk scoring
- Connected quality modules: CAPA, complaints, audits, supplier management
- Real-time visibility across all document statuses and approval queues
Best For: Food manufacturers and CPG companies that run their business on Salesforce and want quality management in the same ecosystem.
Pricing: Custom. Contact ComplianceQuest for pricing.
7. Manufacturo: Best for Shop Floor Document Management
Manufacturo takes a manufacturing-operations-first approach to document management. Rather than starting from compliance frameworks, it starts from the production floor: work instructions, bills of materials, engineering drawings, technical specifications. The platform is designed to ensure that operators always see the current, approved version of a document and never an outdated one.
For food equipment manufacturers or contract manufacturers with heavy technical documentation alongside their food safety records, Manufacturo handles the operational side well. It integrates with ERP, WMS, MES, and PLM systems, making it a good fit for manufacturers who need document control to connect with their production data ecosystem.
The gap for food manufacturers primarily focused on safety certification is that Manufacturo is not built around GFSI scheme structures. It is a strong technical document management platform, but food safety-specific compliance mapping sits outside its core design.
Key Features:
- Version-controlled document storage for SOPs, work instructions, and technical specs
- Workflow automation for routing documents to the right approver at each step
- Shop floor access controls so operators see only current, approved versions
- Integration with ERP, WMS, MES, and PLM systems
- Change management module linked to document updates
Best For: Food equipment manufacturers or contract manufacturers with heavy technical documentation needs such as engineering drawings, specifications, and BOM-linked work instructions.
Pricing: Custom. Contact Manufacturo for a demo and pricing.
8. DocuWare: Best for Document Archiving and Workflow Automation
DocuWare is a mature document management platform with a strong reputation for secure archiving and automated business workflows. It is available as cloud or on-premise deployment, which appeals to manufacturers with specific data residency requirements, and it has a particularly strong presence in European markets.
The platform does centralised document storage, intelligent indexing, and automated routing well. Electronic signatures and access-controlled document sharing are included. Where DocuWare differs from food-safety-native platforms is in what it does not do out of the box: it will not map your documents to SQF clause 2.2.3, it will not flag when a procedure no longer meets a BRCGS Issue 9 requirement, and it does not connect a document revision to a mandatory training sign-off.
For food manufacturers who primarily need a secure, searchable archive with automated routing and who have a separate QMS or compliance tool handling scheme-specific requirements, DocuWare is a solid option.
Key Features:
- Centralised, secure document repository with intelligent indexing
- Automated approval and routing workflows for any document type
- Cloud and on-premise deployment options
- Electronic signatures and access-controlled document sharing
- Integration with Microsoft 365, SAP, QuickBooks, and other platforms
Best For: Food manufacturers who primarily need a secure, searchable document archive with automated routing and who do not require food-certification-specific compliance mapping out of the box.
Pricing: Custom. DocuWare pricing varies by deployment model and user count.
How Document Control Software Supports Food Safety Compliance
Every major food safety certification scheme requires a documented document control system. The specifics vary by scheme, but the core requirements are consistent: documents must be approved before use, version-controlled, accessible to the right people, and removed from circulation when obsolete. Here is how that plays out across the schemes your facility is most likely operating under.
Document Control Under FSMA (Preventive Controls for Human Food)
FDA's FSMA Preventive Controls rule requires covered facilities to maintain a written food safety plan that includes a documented hazard analysis, preventive controls procedures, monitoring procedures, corrective action records, and verification activities. All of these documents must be retained, version-controlled, and retrievable on demand.
Critically, FSMA requires that records be available for FDA review within 24 hours during an inspection. A document management system that cannot produce an audit-ready record on short notice does not meet that requirement. Document control software with timestamped version histories, approval records, and document-type search functionality makes compliance retrieval straightforward rather than stressful.
For food manufacturers looking at the broader FSMA framework, see our guides on FSMA 204 and FSMA compliance software. You can also access FDA's full FSMA rules and guidance for industry directly.
Document Control Requirements in SQF
SQF Code Edition 10, Section 2.2 (Document Control and Records) specifies exactly how documents must be managed at an SQF-certified facility. Documents must be uniquely identified, approved before use, dated, and maintained in a way that prevents unauthorised changes. Obsolete documents must be removed from all points of use and clearly marked if retained for reference.
SQF auditors treat document control as a standalone system element, not background paperwork. They want to see the approval record, the version history, and evidence that staff had access to current documents at the point of use. SQFI's document control guidance for food manufacturers and SQF Tip Sheet 9: Document Control and Records both lay out exactly what an auditor expects to find.
For a deeper look at SQF-specific tools and audit preparation, see our SQF software and SQF audit checklist guides.
BRCGS Issue 9 Document Control Requirements
The BRCGS Global Standard for Food Safety Issue 9 includes document control requirements under clause 3.3. Sites must maintain a documented control system covering document approval, version control, access control, and defined record retention periods.
BRCGS auditors pay particular attention to whether obsolete versions are genuinely inaccessible. Finding a printed SOP on the production floor that is two versions out of date is an immediate non-conformance. Document control software that enforces digital-only distribution and automatically archives superseded versions removes that risk entirely.
For BRCGS-specific audit preparation guidance, see our BRC audit software guide.
FSSC 22000 Version 6 Documentation Requirements
FSSC 22000 version 6 aligns with ISO 22000:2018, meaning its documentation requirements follow ISO's framework for "control of documented information" under clause 7.5. This covers the creation and update of documented information, control of documented information (availability, suitability, protection), and the retention of documented information as evidence of conformity.
In practical terms, FSSC 22000 v6 requires that your food safety management system documentation be current, protected from unintended alteration, and available to the right people at the time of need. Version control, access control, and audit trails are built into the standard itself, not optional add-ons.
Why Food Manufacturers Need More Than a Generic DMS
SharePoint can store your SOPs. Google Drive can organise your folders. DocuWare can archive and route your documents. None of them know what SQF clause 2.2.3 requires, and none of them will tell you when a procedure you uploaded last year no longer meets a scheme update.
Food-safety-native document control software closes the gap between document storage and compliance management. It connects document revisions to mandatory training sign-offs, maps procedures to specific scheme clauses, and surfaces compliance gaps before an auditor does. For facilities managing HACCP compliance software requirements or running under multiple GFSI schemes simultaneously, that distinction matters considerably.
Allera's document control software is built around these exact requirements.
FAQs
What documents need to be controlled in food manufacturing?
The list is longer than most manufacturers expect.
Core document types include SOPs, GMP procedures, HACCP plans and supporting documentation, food safety plans, supplier approval records, and audit documentation.
How is document control software different from a regular document management system?
A document management system handles storage and retrieval.
Document control adds enforced governance: version numbers are locked, approvals are required before distribution, recipients are tracked, and obsolete versions are removed from circulation.
In a regulated food manufacturing environment, that governance layer is what turns a file repository into a compliance tool.
SharePoint or Google Drive can store your SOPs, but they cannot enforce that an outdated version is no longer accessible to production staff.
What is document control software for manufacturing?
Document control software is a centralised system that manages the creation, review, approval, versioning, distribution, and retirement of documents across a manufacturing facility.
For food manufacturers, those documents include SOPs, GMP procedures, HACCP plans, food safety plans, supplier approval records, and audit documentation.
The critical distinction is between storing a document and controlling it: document control enforces a managed lifecycle so that only approved, current versions are in active use at any point.
%20(1).png)

.avif)







.avif)

.avif)