

7 Best Supplier Quality Management Tools For FSQA
.avif)
Supplier quality failures are one of the top causes of food safety incidents and GFSI audit non-conformances. The right software turns a manual, document-heavy burden into an automated compliance system that keeps your team audit-ready year-round.
Most supplier quality management tool listicles are written for pharma and general manufacturing teams. They are not written for food QA managers juggling SQF Edition 10, BRCGS Issue 9, FSMA FSVP requirements, allergen declarations, and COA review across dozens of ingredient suppliers. This list is different. Every tool here is evaluated specifically against food industry workflows and compliance requirements.
The Shortlist
- Allera — Best for food manufacturers: supplier onboarding, compliance tracking, and GFSI audit readiness
- TraceGains — Best for COA and specification management in food and beverage
- Safefood 360° — Best for GFSI-aligned food safety management with built-in supplier monitoring
- Octave Reliance — Best for enterprise-level supplier quality workflows
- MasterControl — Best for regulated industry supplier qualification and document control
- SafetyCulture — Best for mobile-first supplier audits and inspection management
- Kodiak Hub — Best for supplier scorecarding and performance analytics
How We Ranked These Tools
Every tool on this list was evaluated against six criteria:
- Food Industry Fit: Is the platform designed for food manufacturers, or does it require configuration to fit food workflows?
- Compliance Support: Does it natively support GFSI scheme requirements, FSMA recordkeeping, and food-specific document types?
- Ease of Use: Can your QA team and suppliers adopt it without dedicated IT resources?
- Supplier Onboarding: How easy is it for suppliers to submit documents and complete required tasks?
- Scalability: Can the platform grow with your supplier base from 20 to 200 suppliers?
Rankings are based on public vendor documentation, publicly available product information, and published user reviews. No vendor paid to be included or ranked. Allera is ranked #1; we have explained exactly why and included limitations. Readers can judge the reasoning.
#1 Allera
Website: Supplier Management Module
Best for: Small to mid-sized food manufacturers, co-packers, and distributors
Food industry focus: Yes, purpose-built for food safety and quality compliance
Implementation: ~30 days
Why Allera Is #1 for Food Manufacturers
Allera is the only tool on this list built exclusively for food safety and supplier quality management. Every feature is designed around what food QA managers actually need: GFSI-aligned supplier approval workflows, automated document collection, COA tracking, and expiration alerts, without the configuration overhead of a general enterprise QMS.
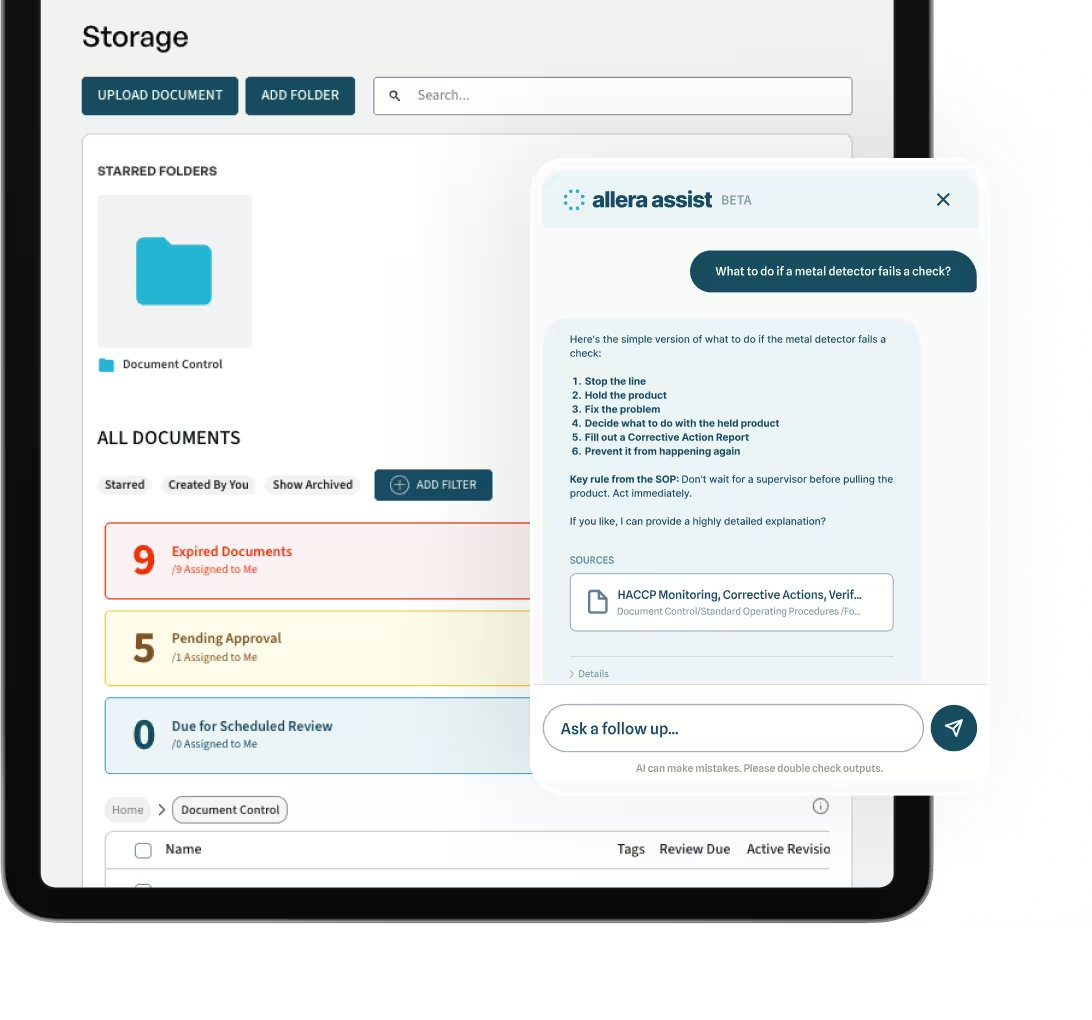
Allera's MagicLink feature is particularly useful for onboarding. Suppliers upload documents and complete questionnaires through a secure link with no account creation required. That removes the friction that typically stalls supplier onboarding programs and generates the email chains that QA teams spend hours managing.
Expiration tracking is automated across all supplier documents and certifications. When a certificate approaches its expiration date, both the supplier and your QA team receive proactive alerts. This closes the most common gap in BRCGS and SQF supplier programs: expired documents that were not caught until an auditor asked for them.
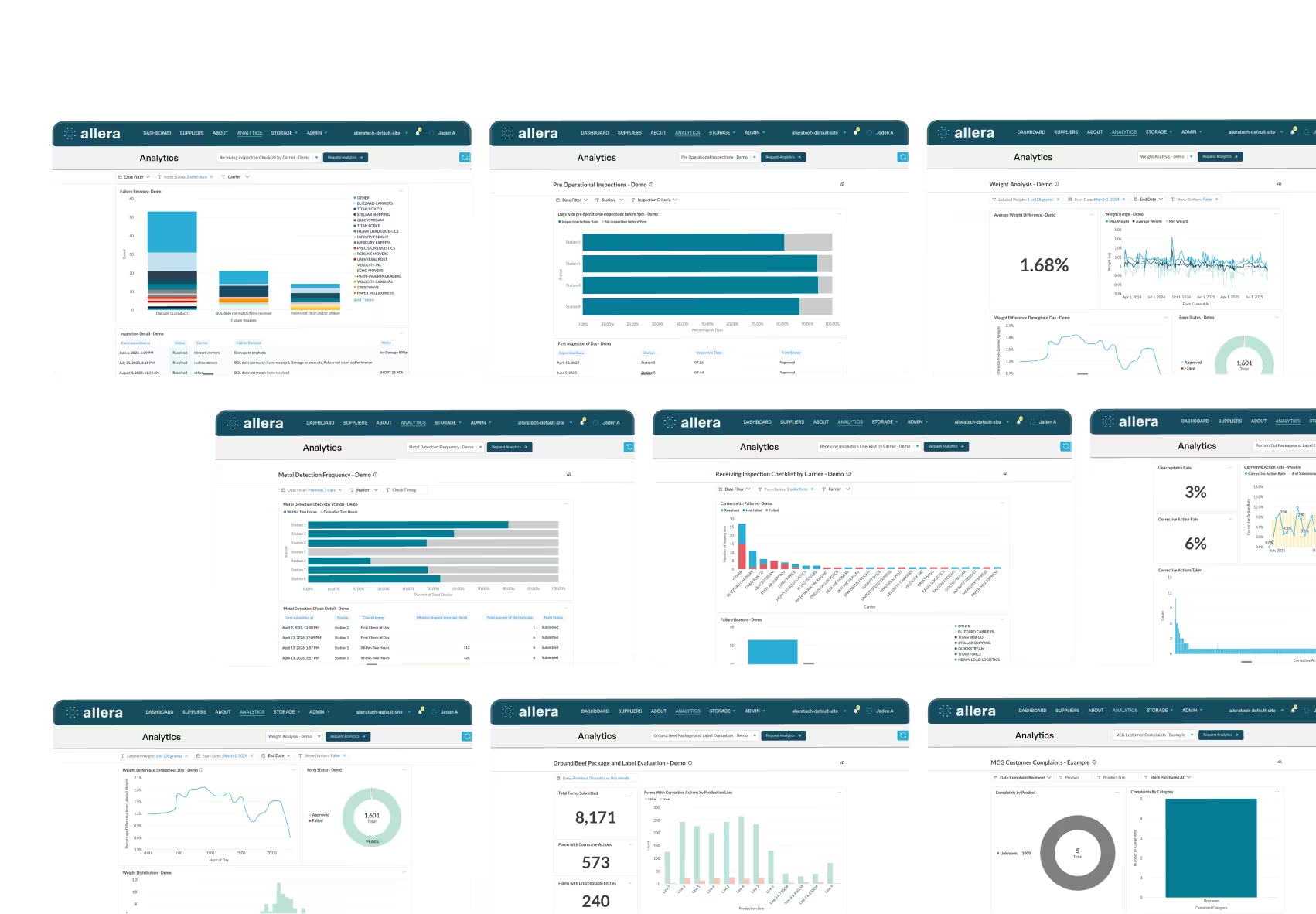
The compliance dashboard gives you real-time visibility into submitted versus outstanding documents by supplier, site, and document type. You can see your audit readiness at a glance rather than building a status report from a spreadsheet before every certification visit.
Allera Key Features
- Supplier onboarding with automated document requests and custom requirements per supplier
- Document collection via MagicLink — suppliers upload without creating an account
- Certificate and document expiration tracking with automated alerts to your QA team and suppliers
- Supplier scorecards tracking compliance rates and supply chain risk metrics
- Supply chain visibility with supplier mapping and risk assessment
- Audit management with scheduling, checklist management, and corrective action follow-up
- Compliance monitoring with real-time status tracking across your supplier network
Customer Proof
Joy Webb, QA Manager at Volpi Foods: "Allera is really effective for managing suppliers and sending out notifications. Whenever we had questions, Jesse was there to help us right away."
See how Allera automates supplier document collection, certificate expiration tracking, and audit readiness for food manufacturers, without spreadsheets or email chains. Explore Allera's supplier management module.
#2 TraceGains
Website: tracegains.com
Best for: Mid to large food and beverage manufacturers
Food industry focus: Built for food and beverage specifically.
Implementation: 2–6 months
What TraceGains Does
TraceGains centralizes supplier documents, COAs, and product specifications in a shared network where suppliers upload documents once and share them across multiple buyers. The platform connects ingredient-level compliance data to finished-goods traceability, which is useful for manufacturers managing complex formulations and multi-supplier ingredient sourcing.
Supplier onboarding, compliance workflows, and audit readiness are built around food and beverage use cases. The network model reduces onboarding friction for suppliers already on the TraceGains platform, since their documents are already in the system and can be shared to new customers without re-uploading.
TraceGains also supports FSMA 204 traceability record keeping, which is increasingly important for food manufacturers managing Key Data Elements and Critical Tracking Events under the FDA's food traceability rule.
TraceGains Key Features
- Networked COA and specification management across the TraceGains supplier community
- Ingredient and finished-goods traceability linked across the supply chain
- Recipe, allergen, and nutrition claims management
- Supplier approval and ongoing monitoring workflows
- FSMA 204 traceability recordkeeping support
- Supplier scorecards, performance tracking, and risk monitoring
- Built-in audit readiness and regulatory reporting
#3 Safefood 360°
Website: safefood360.com
Best for: Medium to large food, ingredient, or beverage manufacturers
Food industry focus: Yes, built for food safety management
Implementation: 3–6 months
What Safefood 360° Does
Safefood 360° is an end-to-end food safety management system that digitizes HACCP plans, CAPA workflows, nonconformance tracking, and supplier management in one platform. It is designed for multi-site food operations that need centralized compliance dashboards and consistent audit documentation across facilities.
The platform supports GFSI-aligned certification schemes including SQF, BRCGS, FSSC 22000 Version 6, and ISO 22000:2018, with supplier monitoring built as part of the broader food safety management system. For multi-site manufacturers, the ability to manage supplier programs and HACCP documentation in the same platform reduces duplication and audit preparation time.
Safefood 360° Key Features
- HACCP planning and CCP monitoring with digital records
- CAPA workflows, nonconformance tracking, and root cause analysis
- Internal audits, verification, and validation management
- Supplier approval and ongoing monitoring for multi-site operations
- Centralized compliance dashboards for inspection and certification audits
- Document management for food safety records and quality documentation
#4 Octave Reliance
Website: octave.com
Best for: Enterprise food and beverage manufacturers (multi-site, global)
Food industry focus: Moderate, general enterprise QMS with food modules
Implementation: 6–12 months
What Octave Reliance Does
ETQ Reliance is an enterprise quality management system used across food, pharma, and industrial sectors. It manages supplier quality, document control, CAPA, and audit workflows at scale, with highly configurable modules that can be adapted to SQF, BRCGS, and FSMA compliance requirements.
For global food manufacturers with complex regulatory environments and multi-facility operations, Octave provides configuration depth and scalability that purpose-built food safety tools often cannot match. The tradeoff is implementation time and cost. A 6–12 month implementation is typical, and the platform requires dedicated IT and QMS administration resources to configure and maintain.
Octave is not a practical choice for small or mid-sized food companies. For enterprise manufacturers managing hundreds of suppliers across multiple countries, the configurability and ERP integration depth justify the investment.
Octave Reliance Key Features
- Enterprise QMS for food and beverage companies with configurable supplier quality modules
- Document control with versioning and access control across facilities
- Supplier quality management with corrective action tracking
- Audit management with automated workflows, scheduling, and follow-up
- Risk assessment and change management across global operations
- Configurable workflows that scale across multiple plants and sites
- Integration with ERP systems for unified quality data
#5 MasterControl
Website: mastercontrol.com
Best for: Regulated industry manufacturers including food, pharma, and medical device
Food industry focus: Moderate, broader regulated-industry platform
Implementation: 3–9 months
What MasterControl Does
MasterControl is a comprehensive QMS platform built for regulated manufacturers who need strict documentation control and audit trails. It centralizes supplier onboarding, qualification, audits, and corrective actions, tying supplier documents directly to product and quality requirements throughout the supply chain.
The platform's strength is electronic records management and document control designed for FDA-regulated environments, including electronic signatures and 21 CFR Part 11 compliance. For food manufacturers operating in regulated ingredient categories, or those that also supply pharmaceutical or nutraceutical markets, MasterControl provides a unified quality and supplier compliance system.
MasterControl is not built around food industry certifications like SQF or BRCGS natively. Teams with food scheme audit requirements will need to configure the platform to match those workflows, which adds implementation time and requires more internal administration than a purpose-built food tool.
MasterControl Key Features
- Supplier document control and qualification workflows
- Supplier audit scheduling and findings management
- CAPA tracking tied to supplier non-conformances
- Document control with electronic signature and 21 CFR Part 11 compliance
- Training management and SOP control linked to supplier programs
- Reporting and analytics across supplier performance metrics
#6 SafetyCulture
- Website: safetyculture.com
- Best for: Operations and QA teams needing mobile-first audit and inspection tools
- Food industry focus: Moderate, widely used in food manufacturing for GMP and supplier audits
- Implementation: Days to weeks
What SafetyCulture Does
SafetyCulture is an operations management platform built around mobile-first audits, inspections, and checklists. It is widely used by food manufacturers for GMP inspections, supplier audits, and corrective action management. The template library includes food safety inspection and audit checklists, and the mobile experience makes it practical for frontline teams and supplier facility visits.
Where SafetyCulture fits well is audit execution. Capturing findings, assigning corrective actions, and tracking completion are all straightforward on mobile. Where it falls short as a complete supplier quality system is document collection, certificate tracking, and FSVP recordkeeping. These require manual workarounds that are not sustainable at scale across a large supplier base.
If you need a mobile audit tool to complement an existing supplier management system, SafetyCulture is a strong choice for that specific use case. If you need a complete supplier quality management platform with automated document collection and expiration tracking, you will need a dedicated solution.
SafetyCulture Key Features
- Mobile auditing and inspection execution with offline capability
- Customizable checklists and audit templates including food safety formats
- Corrective action management with photo capture and assignment
- Real-time reporting and performance dashboards
- Integration with QMS and ERP systems
- Training management and document sharing
#7 Kodiak Hub
- Website: kodiakhub.com
- Best for: Mid to large manufacturers needing supplier scorecarding and performance analytics
- Food industry focus: Low-moderate, general supplier relationship management
- Implementation: 1–3 months
What Kodiak Hub Does
Kodiak Hub is a supplier relationship management platform focused on performance scorecarding, risk monitoring, and supplier collaboration. It tracks supplier sustainability and ESG metrics alongside quality and compliance data, making it a good fit for procurement and supply chain teams managing a broad supplier portfolio.
For food manufacturers whose primary need is regulatory compliance and food safety document management, Kodiak Hub is a partial fit. The platform does not have native support for SQF, BRCGS, or FSSC 22000 supplier requirements, and FSVP workflows require custom configuration. COA management and allergen document collection are not built-in features.
Where Kodiak Hub earns its place on this list is supplier performance analytics. If you are managing a large supplier base and need robust scorecarding, AI-powered risk monitoring, and supplier development tools, the platform delivers strong analytics capabilities that food-specific tools often do not match.
Kodiak Hub Key Features
- Dynamic supplier scorecards with real-time performance metrics
- ESG, sustainability, and risk tracking capabilities
- Corrective action workflows for supplier noncompliance
- Data integrations to unify supplier and compliance data from multiple sources
- Sourcing strategy and supplier development tools
- AI-powered supplier risk monitoring
- Supplier onboarding and collaboration portal
What to Look for in Supplier Quality Management Tools
Food manufacturers have different needs than pharma or industrial buyers. If you are managing ingredient suppliers, you need tools that support GFSI scheme certification, FSMA legal requirements, and the document types that food auditors actually check. Here is what matters most.
GFSI Scheme Alignment (SQF, BRCGS, FSSC 22000)
If your facility is SQF, BRCGS, or FSSC 22000 certified, your supplier management program is not optional. It is a scheme requirement. SQF Edition 10, BRCGS Issue 9, and FSSC 22000 all mandate documented supplier approval programs and ongoing monitoring processes.
That means your software needs to support the specific document types, audit frequencies, and risk-based monitoring protocols those schemes require. The GFSI Benchmarking Requirements 2024 sets the baseline for what certified schemes must cover. When evaluating tools, ask vendors directly whether the platform is designed around GFSI requirements or whether alignment requires custom configuration.
FSMA Compliance and Foreign Supplier Verification (FSVP)
If you import ingredients from outside the United States, you are likely subject to the FDA FSMA Foreign Supplier Verification Program (FSVP). FSVP requires you to conduct and document risk-based verification activities for each foreign supplier, including hazard analysis and ongoing verification records.
Even if you do not import directly, the FDA FSMA Preventive Controls Supply-Chain Program requires supply-chain preventive controls for hazards your suppliers are controlling. Your supplier quality management software needs to support the scheduling, recordkeeping, and documentation requirements for both programs.
COA and Specification Management
Certificates of Analysis are among the most reviewed documents during GFSI and FSMA audits. For high-risk ingredients, you need COA review per lot, with out-of-spec flagging before the material enters your production line.
Good supplier quality software automates COA collection, routes documents for review, and flags values that fall outside your approved specifications. Without automation, COA management becomes a manual process that breaks down quickly as your supplier count grows.
Supplier Onboarding and Document Collection
Getting suppliers to submit the right documents is often the hardest part of running a supplier approval program. You need certifications, allergen declarations, specifications, and questionnaire responses from every supplier, before they start shipping you ingredients.
The best tools eliminate email chains by giving suppliers a secure, direct way to upload documents and complete onboarding tasks. Allera's food supplier management software includes MagicLink, which lets suppliers submit documents without creating an account, removing the most common barrier to supplier onboarding.
Certificate Expiration Tracking and Alerts
Expired third-party certificates are one of the most common findings in SQF and BRCGS certification audits. If your supplier's GFSI certificate expires and you do not catch it before your audit, you have a non-conformance.
Your supplier quality software should automatically track expiration dates and alert both your QA team and the supplier before the deadline. Manual calendar reminders do not scale across 30, 50, or 100 suppliers. The SQF Institute guidance documents and BRCGS Issue 9 both require evidence of ongoing supplier monitoring, which depends on keeping certification records current.
Audit Management and CAPA Workflows
Supplier audits generate findings that need to be tracked and closed. Good supplier quality tools let you schedule audits, capture findings on-site, assign corrective actions, and track closure through to verification, all within a single system.
Separate spreadsheets for scheduling, findings, and CAPA closure are a common source of audit preparation pain. A unified system makes it straightforward to demonstrate corrective action closure during your food safety audit without pulling records from multiple places.
Supplier Scorecarding and Performance Dashboards
Over time, you need data to justify supplier approvals, identify underperforming suppliers, and prioritize your QA team's attention. Supplier scorecards let you track defect rates, on-time document submission, audit performance, and compliance rates across your entire supplier base.
Real-time dashboards eliminate the need to run reports manually each quarter. They also make it straightforward to present supplier performance data to leadership or to auditors when they ask how you monitor approved suppliers.
If GFSI scheme alignment and FSMA readiness are your top priorities, Allera is built for exactly that. See how food manufacturers use it to get audit-ready faster.
FAQs
What is the difference between supplier quality management and supplier relationship management?
Supplier quality management (SQM) focuses on compliance, documentation, and food safety requirements. It covers supplier approval, document collection, audit management, corrective actions, and regulatory recordkeeping.
Supplier relationship management (SRM) focuses on the broader commercial and strategic relationship with suppliers, including performance scorecarding, sourcing strategy, ESG metrics, and supplier development. Kodiak Hub leans toward SRM. Allera and TraceGains are purpose-built for SQM in food manufacturing contexts. Most food QA managers should prioritize SQM capabilities if their primary driver is certification compliance and FSMA readiness.
How do I choose between a food-specific tool and a general QMS platform?
Start with your primary use case. If your team needs a dedicated supplier compliance platform with fast implementation and food-specific workflows, a purpose-built tool like Allera or TraceGains will get you operational faster with less configuration overhead.
If your facility needs an integrated QMS covering HACCP, internal audits, document control, and supplier management in one enterprise system, a broader platform like ETQ or MasterControl may justify the longer implementation and higher cost. Company size and IT resources matter too. Enterprise platforms require more administration; purpose-built tools are designed for smaller QA teams to manage independently.
What documents should supplier quality management tools collect from food suppliers?
Standard documents to collect from food suppliers include:
- Approved supplier certificates (GFSI, organic, kosher, halal where applicable)
- Product specifications
- Allergen declarations
- Certificates of analysis per lot or shipment
- HACCP plans for high-risk suppliers
- Completed supplier questionnaires
The USDA FSIS HACCP compliance guidance provides a reference for what HACCP documentation should cover. For imported ingredients, you may also need third-party audit reports and hazard analysis records for FSVP compliance. Your certification scheme requirements will define additional documentation requirements specific to SQF, BRCGS, or FSSC 22000.
How is supplier quality management software different from a QMS?
A quality management system (QMS) covers the full scope of your facility's quality and food safety programs, including HACCP, internal audits, document control, calibration, and training. Supplier quality management software focuses specifically on managing your supplier relationships, approvals, and compliance documentation.
Some platforms like Allera focus exclusively on supplier management and document control, while others like ETQ Reliance and MasterControl bundle supplier management as one module within a broader enterprise QMS.
What should supplier quality management tools track for food safety compliance?
At minimum, your supplier quality management tools should track:
- Supplier approval status
- Required certifications and their expiration dates
- Allergen declarations
- Product specifications
- COAs per lot for high-risk ingredients
- Audit schedules and findings
- Corrective action closure
For FSMA-regulated facilities, FSVP records and supply-chain preventive control documentation are also required. For GFSI-certified facilities, the tool should support the specific monitoring frequency and documentation requirements of your certification scheme, whether SQF, BRCGS, or FSSC 22000.
What is FSMA FSVP and how does supplier management software help with it?
FSMA FSVP (Foreign Supplier Verification Program) is an FDA regulation requiring importers of food to conduct risk-based verification activities for each foreign supplier. Required activities include hazard analysis, supplier performance evaluation, and documented verification records.
Supplier quality management software helps by automating recordkeeping, scheduling required verification activities, and maintaining the audit trails that FDA inspectors expect to see. Without a dedicated system, FSVP compliance becomes a significant manual recordkeeping burden that is difficult to sustain consistently.
Do I need supplier quality management tools if I am SQF or BRCGS certified?
Yes. SQF Edition 10 and BRCGS Issue 9 both require documented supplier approval programs with ongoing monitoring and periodic re-evaluation. These are not optional elements of the standard.
Auditors will specifically review your supplier qualification records, approval documentation, and evidence of ongoing monitoring during your food safety audit. Software makes it much easier to demonstrate compliance than a manual spreadsheet or folder system, particularly when auditors request records on short notice.
What is the best supplier quality management software for food manufacturers?
The best supplier quality management software for food manufacturers is one built specifically for food industry workflows. Allera is purpose-built for food safety and supplier compliance, with native support for GFSI scheme requirements, automated COA and certificate tracking, and a passwordless supplier portal that simplifies onboarding.
For larger food and beverage companies with complex formulations and supply chains, TraceGains is the strongest alternative for COA and specification management at scale.
What does supplier quality management include?
Supplier quality management covers the full lifecycle of managing your supplier relationships from a quality and compliance perspective. This includes:
- Initial supplier approval and qualification
- Document collection (certificates, specifications, allergen declarations, COAs)
- Ongoing performance monitoring
- Scheduled audits and inspections
- Corrective action management
- Annual reviews
For food manufacturers, it also includes GFSI scheme alignment and FSMA recordkeeping as legally required components of your supplier program.
What is supplier quality management software?
Supplier quality management software is a digital platform that helps food manufacturers and other regulated businesses manage their supplier approval, monitoring, and compliance programs. It replaces manual spreadsheets and email-based document collection with automated workflows for onboarding suppliers, collecting required documents, tracking compliance status, managing audits, and issuing corrective actions.
For food manufacturers, it also handles the GFSI-specific documentation and FSMA recordkeeping requirements that general tools often miss.
%20(1).png)

.avif)







.avif)
.avif)
.avif)

.avif)

%20(1).avif)


.avif)
.avif)



.avif)


